- The root-mean-square value of an alternating current is equivalent to the steady direct current that would dissipate heat at the same rate as the alternating current in a given resistor.
- For a sinusoidal source,
(a) the root mean square value of the current is given by $$I_{rms}=\frac{I_o}{\sqrt{2}}$$.
(b) the mean or average power < P > absorbed by a resistive load is half the maximum power.
$$<P>=\frac{1}{2}P_o=\frac{1}{2}{I_o}{V_o}=\frac{1}{2}{I_o}^2R =\frac{V_o^2}{2R}$$.
- An a.c. transformer is a device for increasing or decreasing an a.c. voltage. It consists of a primary coil of Np turns and voltage Vp and secondary coil of Ns turns and voltage Vs wrapped around an iron core.
- For an ideal transformer (assuming no energy is lost), the following equation is obeyed
$$\frac{N_s}{N_p}=\frac{V_s}{V_p}=\frac{I_p}{I_s}$$. - Power loss in the transmission lines is minimized if the power is transmitted at high voltages (i.e. low currents) since $$P_{loss}=I^2R$$ where I is the current through the cables and R is the resistance of the cables.
- The equation $$P=\frac{V^2}{R}$$ is often mistakenly used to suggest that power lost is high when voltage of transmission is high. In fact, V refers to the potential difference across the cables, which often have but a fraction of the overall resistance through which the current passes.
[accordions autoHeight=’true’]
[accordion title=”1. Particle Nature of Light”]
- A photon is a quantum of electromagnetic radiation.
- The energy of a photon is given by E=hf, where h is Planck’s constant (6.63 $$\times$$ 10-34 J s) and f is its frequency.
[/accordion]
[accordion title=”1.1 Photoelectric Effect”]
- The photoelectric effect is the emission of electrons from a metal surface when electromagnetic radiation of sufficiently high frequency is shone on it.
- The energy of an incident photon is the sum of the maximum kinetic energy $$K.E._{max}$$ of the emitted electrons from the metal surface and the work function $$\Phi$$ of the metal. Einstein’s photoelectric equation states that
$$hf=\Phi +K.E._{max}=hf_o +K.E._{max}$$
- where $$f_o$$ is the threshold frequency or minimum frequency of the electromagnetic radiation below which no electrons are emitted from the metal surface regardless of the intensity of the radiation.
- The work function $$\Phi$$ of a metal is the minimum energy needed to remove an electron from the metal surface.
- $$K.E._{max}$$ can be measured by applying a voltage to prevent the emitted electrons from reaching the electrode that collects them. This voltage is known as the stopping voltage $$V_s$$ and since the charge of an electron is e, the equation can be rewritten as
$$hf=\Phi + eV_s$$.
[/accordion]
[accordion title=”1.2 Line Spectra”]
- An atom is in the ground state when its electron occupies the lowest energy level. When the atom gains energy, its ground state electron makes a transition to a higher energy level. The atom is said to be in an excited state.
- At this excited state, the electron is unstable. It will jump to a lower energy level by emitting a photon whose energy is equal to the energy difference between the two levels. The photon energy is given hf = Ehigher – Elower.
- The emission line spectra are the spectra of light radiated by individual atoms in a hot gas when the electrons in the atoms jump from higher energy levels to lower energy levels. Each spectrum consists of coloured lines on a dark background.
- The absorption line spectra consists of dark lines on a coloured background. When a beam of white light is passed through a cool gas, photons whose energies are equal to the excitation energies of the gas atoms, are absorbed. These photons are re-emitted in all directions, so the intensity of these wavelengths in the transmitted white light beam is reduced.
[/accordion]
[accordion title=”2. Wave Nature of Particles”]
- Louis de Broglie postulated that, because photons have wave and particle characteristics, perhaps all forms of matter have both properties. Electron diffraction provides evidence for the wave nature of particles.
- The de Broglie wavelength of a particle is given by $$\lambda = \dfrac{h}{p}$$ where p is the momentum (mv) of the particle and h is Planck’s constant.
[/accordion]
[accordion title=”3. X-ray Spectrum”]
[/accordion]
[accordion title=”4. Heisenberg Uncertainty Principle”]
[/accordion]
[accordion title=”5. Wave Function and Probability”]
- An electron can be described by a wave function $$\Psi$$ where the square of the amplitude of the wave function $$|{\Psi}|^2$$ gives the probability of finding the electron at a point.
[/accordion]
[accordion title=”6. Quantum Tunneling”]
- Classically, an electron of energy E approaching a potential barrier, whose height U is greater than E, cannot penetrate the barrier but would simply be reflected and return in the opposite direction.
- However, quantum mechanics predicts that since $$|{\Psi}^2|$$ is non-zero beyond the barrier, there is a finite chance of this electron tunnelling through the barrier and reaching the other side of the barrier.
- The transmission coefficient T represents the probability with which an approaching electron will penetrate to the other side of the barrier. The transmission coefficient T is given by $$T=e^{-2kd}$$ where $$k=\sqrt{\dfrac{8\pi^2m(U-E)}{h^2}}$$
[/accordion]
[/accordions]
The Nucleus
- existence and size demonstrated using the Rutherford $$\alpha$$-scattering experiment.
- consists of nucleons (protons and neutrons)
- isotopes of an element share the same number of protons but different number of neutrons.
Nuclear Reactions
- nuclear reactions involve two or more reactants.
- represented using the form: $${^{14}_7N}+{^4_2He}\rightarrow{^{17}_8O}+{^1_1H}$$
- for a reaction that releases energy, mass-energy of reactants = mass-energy of products + E,
where $$E = mc^2$$ and m is the mass defect (difference in mass between the products and reactants). - binding energy is the energy released when the nucleus is formed from its separate protons and neutrons. The same amount of energy is required to break up a nucleus into its constituent nucleons.
- binding energy per nucleon ($$\frac{B.E.}{A}$$) is an indication of the stability of a nucleus, where B.E .is binding energy and A is the nucleon number. You need to know how to sketch its variation with nucleon number. (The following video explains the shape of the $$\frac{B.E.}{A}$$ versus A graph and why it peaks at $$^{56}Fe$$.
- nuclear fission is the disintegration of a heavy nucleus into two lighter nuclei of comparable mass with the emission of neutrons and release of energy.
e.g. $${^{235}_{92}U}+{^1_0n}\rightarrow{^{236}_{92}U}\rightarrow{^{144}_{56}Ba}+{^{90}_{36}Kr}+2^1_0n+Energy$$ - nuclear fusion occurs when two light nuclei combine to form a single more massive nucleus, leading to the release of energy.
e.g. $${^2_1H}+{^3_1H}\rightarrow{^4_2He}+{^1_0n}+Energy$$
- The following quantities are always conserved:
- proton number & neutron number
- momentum
- mass-energy
Radioactive Decay
- spontaneous and random emission of radiation from a radioactive nucleus.
- $$\alpha$$ particle – helium nucleus
- $$\beta$$ particle – electron
- $$\gamma$$ particle – electromagnetic radiation
http://youtu.be/Qlb5Z8QBpcI
- $$A=-\frac{dN}{dt}=\lambda N$$
where A is the rate of disintegration or activity, N is the number of radioactive nuclei and $$\lambda$$ is the decay constant. - $$x=x_0{e^{-\lambda t}}$$
where x could represent the activity, number of undecayed particles or received count rate. - half-life ($$t_{\frac{1}{2}}$$) is the average time taken for half the original number of radioactive nuclei to decay.
- From $$x=x_0{e^{-\lambda t}}$$,
$$\frac{x}{x_0}=\frac{1}{2}=e^{-\lambda t_{\frac{1}{2}}}$$
$$\Rightarrow{-ln2}=-\lambda t_{\frac{1}{2}}$$
$$\Rightarrow{t_{\frac{1}{2}}}=\frac{ln 2}{\lambda}$$ - You may also use $${\frac{x}{x_0}}={\frac{1}{2}}^{\frac{t}{t_{1/2}}}$$, as shown in the following video.
[accordions autoHeight=’true’]
[accordion title=”1. Definitions”]
- The magnetic flux density at a point is defined as the force acting per unit current per unit length of the conductor when the conductor is placed at right angles to the field.
- One tesla is the uniform magnetic flux density which, acting normally to a long straight wire carrying a current of 1 ampere, causes a force per unit length of 1 N m–1 on the conductor.
[/accordion]
[accordion title=”2. Magnetic Fields”]
- The following are the vector symbols used in diagrams to represent the direction of vectors in 3 dimensional space:
- $$\rightarrow$$ : on the plane of the page
- $$\otimes$$ : into of the page
- $$\odot$$ : out of the page
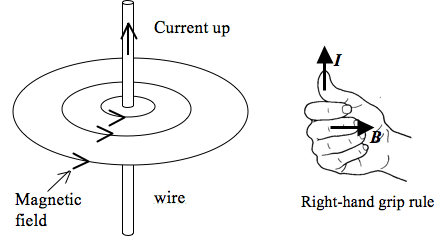
- The following are some important points to take note when representing a magnetic field by magnetic field lines:
- Magnetic field lines appear to originate from the north pole and end on the south pole.
- Magnetic field lines are smooth curves.
- Magnetic field lines never touch or cross.
- The strength of the magnetic field is indicated by the distance between the lines – closer lines mean a stronger field.
[/accordion]
[accordion title=”3. Force on a Current-Carrying Conductor in a Magnetic Field”]
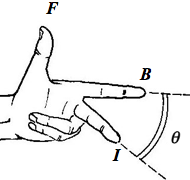
- When a wire of length $$l$$ carrying a current $$I$$ lies in a magnetic field of flux density $$B$$ and the angle between the current $$I$$ and the field lines $$B$$ is $$\theta$$, the magnitude of the force $$F$$ on the conductor is given by $$F = BIl sin \theta$$.
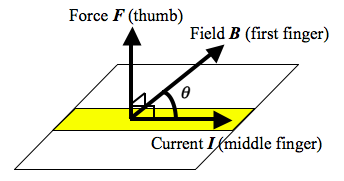
- The directions of the vectors can be recalled by using the Fleming’s Left-Hand Rule.
[/accordion]
[accordion title=”4. Force on a Moving Charge in a Magnetic Field”]
- A charge $$q$$ travelling at constant speed $$v$$ at an angle $$theta$$ to a magnetic field of flux density $$B$$ experiences a force $$F = Bqv sin\theta$$.
[/accordion]
[accordion title=”5. Magnetic fields of current-carrying conductors”]
[/accordion]
[accordion title=”6. Ferromagnetic Materials”]
[/accordion]
[accordion title=”7. Force between Two Parallel Current-Carrying Conductors”]
- Like currents attract and unlike currents repel.
[/accordion]
[/accordions]
Types of Forces
- Static friction
- Frictional force between surfaces at rest with respect to each other.
- It increases with increasing applied force up to a maximum value (which is proportional to normal contact force).
- Kinetic friction
- Frictional force acting between surfaces in relative motion.
- Viscous forces
- Resistive force experienced by a solid moving in a fluid.
- Dependent on speed of object v, e.g. $$F_D\propto v$$ at low speeds and $$F_D\propto v^2$$ at high speeds.
[accordions autoHeight='true']
[accordion title=”1. Newton’s Laws of Motion”]
- Newton’s First Law: a body will remain in its state of rest or uniform motion in a straight line unless acted upon by a resultant force.
- Newton’s Second Law: the rate of change of momentum of a body is proportional to the resultant force acting on it and the change takes place in the direction of the resultant force.
- $$F =\frac{dp}{dt}$$ in general
- $$F =ma$$ when mass is constant.
- Newton’s Third Law: if body A exerts a force on body B, then body B exerts an equal and opposite force on body A
[/accordion]
[accordion title=”2. Linear Momentum”]
- The linear momentum of a body is defined as the product of its mass and its velocity.
- Impulse is the product of the force acting on a body and the time interval during which the force is exerted. It is equal to the change in momentum of the body.
- For constant force, impulse = $$\Delta p =F \Delta t$$
- In general, impulse = $$\Delta p =\int {F .dt}$$
[/accordion]
[accordion title=”3. Collision Problems”]
- The principle of conservation of momentum states that the total momentum of a system of colliding objects remains constant provided no resultant external force acts on the system.
- Conservation of momentum applies to both elastic and inelastic collisions.
- $$m_1u_1+m_2u_2=m_1v_1+m_2v_2$$
- Conservation of kinetic energy applies only to elastic collisions.
- $$\frac{1}{2}m_1u_1^2+\frac{1}{2}m_2u_2^2=\frac{1}{2}m_1v_1^2+\frac{1}{2}m_2v_2^2$$
- Relative speed of approach = Relative speed of separation
- $$u_2-u_1=v_1-v_2$$
[/accordion]
[/accordions]